|
4/10/2023 0 Comments Define ion bonding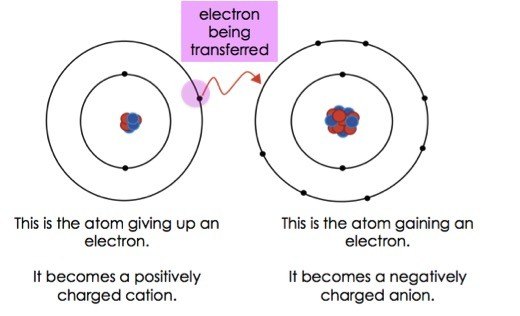
This makes graphite soft and slippery, useful as a lubricant or in pencils, since the layers can separate and slide over each other easily. Chemical bonding is a key concept in chemistry to understand how these elements join together to form various compounds. This will give one atom a positive formal charge and the other a negative formal. The electron moves between layers of carbon atoms which are held together with weak intermolecular forces which are easily broken. There are four types of bonds or interactions: ionic, covalent, hydrogen bonds, and van der Waals interactions. An ionic bond is where one atom takes one valence electron from the other. As the fourth electron in carbon’s outer electron shell is unbonded, it is free to move (delocalised) and can conduct electricity. Dissolved in water, ionic bonds are aqueous, that is, they can conduct. 
Ionic bonds also melt at high temperatures. The structure of the bond is rigid, strong and often crystalline and solid. The bond is typically between a metal and a non-metal. 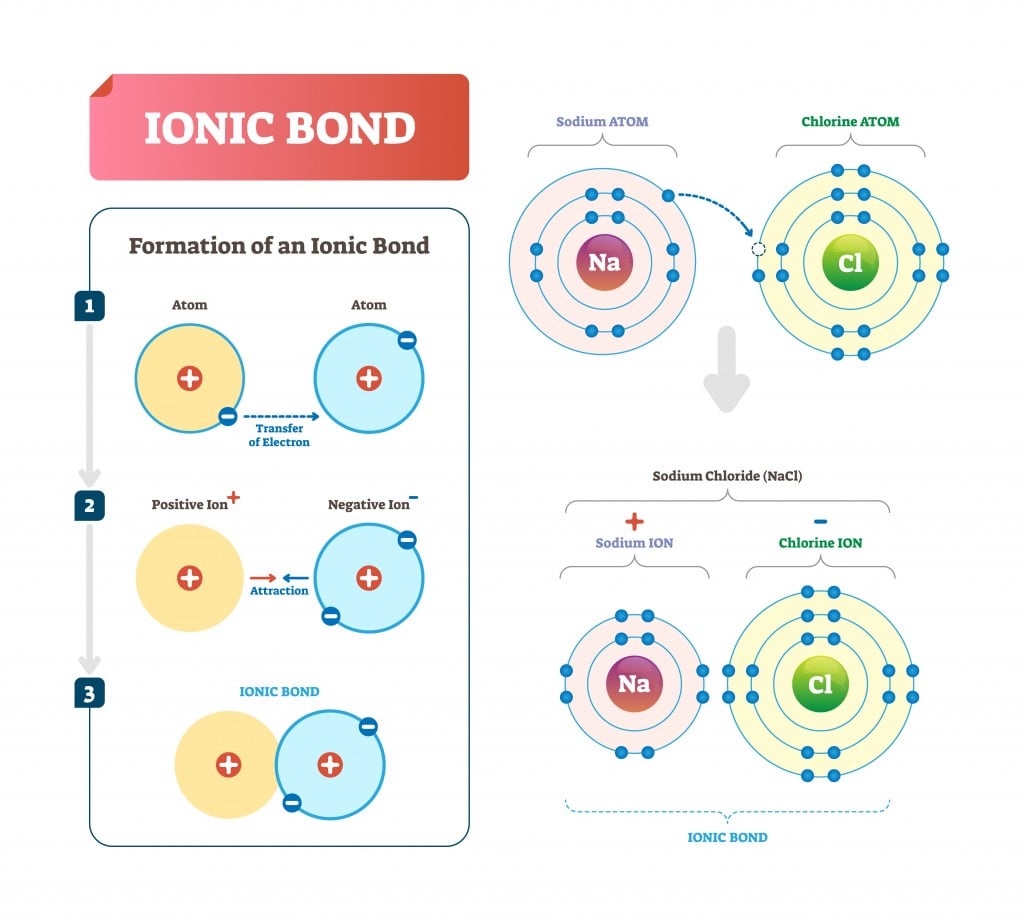
In contrast, each carbon atom in graphite forms only three other bonds. Ionic bonds are atomic bonds created by the attraction of two differently charged ions. In diamond, each carbon atom bonds to four other carbon atoms to form a strong, rigid structure which makes diamonds one of the hardest materials on Earth and virtually unbreakable. An exception is graphite, which has delocalised electrons between layers of carbon atoms.Īlthough diamond and graphite are both giant covalent structures made up of only carbon atoms, they have very different properties due to the way that the carbon atoms are arranged. They have very high melting and boiling points because the strong covalent bonds need to break when these molecules change states.Ĭovalent molecules are not made up of charged particles, so neither simple nor giant covalent structures are able to conduct electricity. Giant covalent structures, such as diamond, graphite and silicon dioxide (silica) are made up of a large number of atoms covalently bonded in a huge network. Heavier molecules have a larger number of electrons which means they can form more intermolecular forces, thereby increasing their melting and boiling point. When the electrons being shared are so unevenly distributed between the atoms the bond that is formed is called an ionic bond. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonding in sodium chloride Examples of Ionic Bonds. The electrostatic attraction between them forms an ionic bond, resulting in a stable ionic compound called sodium chloride (AKA table salt). This is because the weak intermolecular forces that are found between molecules form from electrons. This creates a positively-charged sodium ion and a negatively-charged chlorine ion. Although all simple covalent molecules will have low melting and boiling points, those with a large molecular mass will have a higher melting and boiling point than a molecule with a smaller mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
